Have you ever wondered why some things stick together while others stay apart? In chemistry, atoms are like tiny people with different personalities. Some atoms really love to grab extra electrons, while others do not care at all. This “love” for grabbing an electron is called electron affinity. Understanding the electron affinity trend helps us predict how different elements will behave when they meet each other. It is like having a map that tells you who is hungry for energy and who is full. We are going to explore this map together today in a way that is super easy to follow. You do not need to be a scientist to get this!
What Exactly is Electron Affinity?
Before we dive into the electron affinity trend, let’s define what we are talking about. Imagine an atom is holding a small party. An electron is like a new guest trying to join that party. When the atom takes in that new guest, it usually releases some energy. This energy release is what we call electron affinity. If an atom releases a lot of energy, it means it really wanted that electron. If it doesn’t release much, it wasn’t that interested. Scientists measure this to see how “greedy” an atom is. It is a very important part of how chemicals react to make the things we use every day.
How the Electron Affinity Trend Works
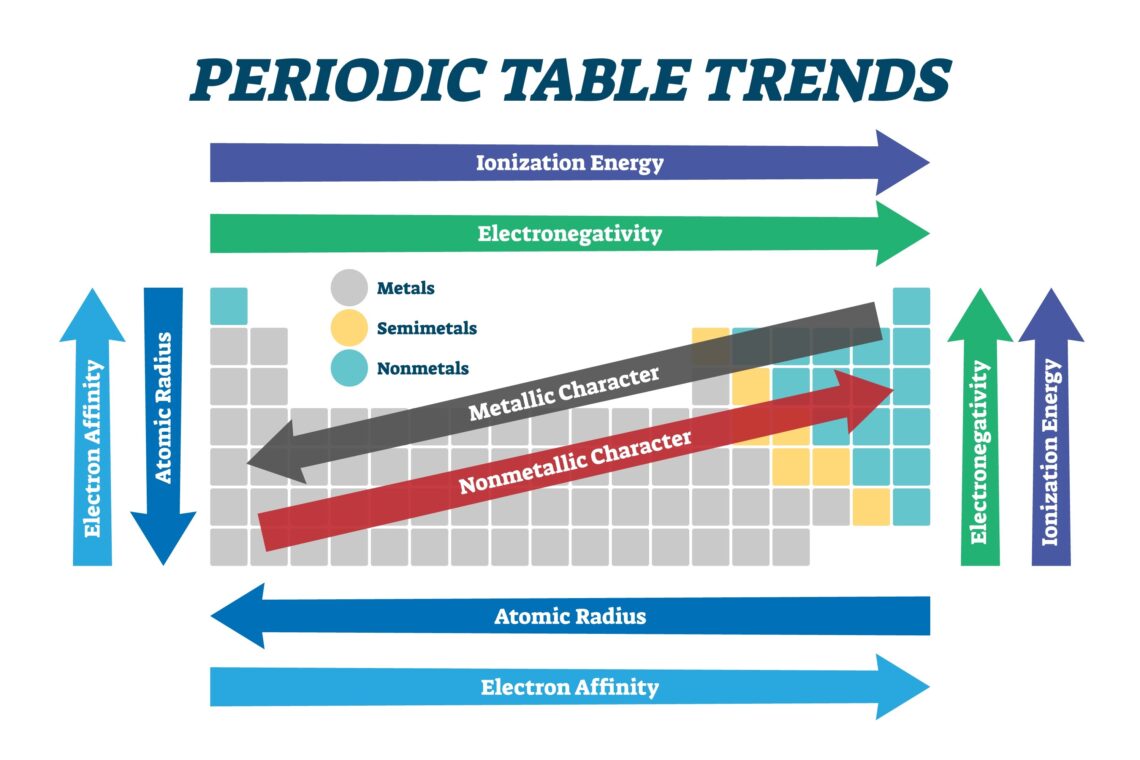
The electron affinity trend is a pattern we see on the Periodic Table of Elements. Just like a staircase or a road, the table has a specific flow. As you move from the left side to the right side, the atoms generally get hungrier for electrons. This happens because the atoms are getting smaller and their centers are getting stronger. A stronger center can pull in a new electron much more easily. This trend is one of the first things students learn because it explains why some elements, like Fluorine, are so reactive. It is all about that pull and the energy that comes with it.
Shutterstock
Moving Across the Periodic Table
When you look at a row on the periodic table, you are seeing the electron affinity trend in action. As you move from left to right, the “hunger” for electrons usually goes up. The atoms on the right side have more protons in their middle. Protons are like magnets for electrons. Because they have more “magnets,” they can snap up a passing electron very quickly. This makes the energy release much higher. By the time you get to the Halogens, the atoms are very excited to grab just one more electron to feel complete. It is a very consistent pattern that helps us organize the elements.
Going Down the Columns
Now, let’s look at what happens when you go from the top to the bottom of the table. The electron affinity trend actually tends to decrease as you go down. This is because the atoms are getting bigger and “fatter.” They have more layers of electrons, which acts like a shield. This shield makes it harder for the center of the atom to feel a new electron on the outside. Imagine trying to hold a ball while wearing ten pairs of thick gloves. It is much harder to grip! That is why the elements at the bottom of a column aren’t as good at grabbing electrons as the ones at the top.
Why Do We Care About This Trend?
You might ask why the electron affinity trend matters to us in real life. Well, this trend explains why some metals rust and why some gases are dangerous. For example, knowing how much an atom wants an electron helps doctors create medicines. It also helps engineers build better batteries for our phones. Every piece of technology we use relies on how electrons move between atoms. If we didn’t understand these trends, we wouldn’t know how to mix chemicals safely. It is the secret code that runs the physical world around us every single day.
The Special Case of Noble Gases
Even though the electron affinity trend usually goes up as you move right, there is a group that breaks the rules. These are the Noble Gases, like Neon and Helium. They are at the very end of the rows. These atoms are already “full” and very happy. They do not want any new guests at their party! Because of this, their electron affinity is actually very low or even zero. They are the calmest elements on the table. Learning about these “rule-breakers” is a fun way to see that chemistry always has some interesting surprises waiting for us.
Metals vs. Non-Metals
The electron affinity trend also shows us the big difference between metals and non-metals. Metals, like Gold or Iron, usually have a low electron affinity. They would actually rather give away electrons than take them! Non-metals, like Oxygen and Chlorine, are the opposite. They have a very high electron affinity. This is why metals and non-metals love to work together. The metal gives, the non-metal takes, and they form a strong bond. This is how we get things like table salt. It is a perfect partnership based on these energy trends.
Factors That Change the Trend
There are a few things that can wiggle the electron affinity trend a little bit. One is the size of the atom, which we mentioned. Another is how the electrons are packed inside. Sometimes, if the inside is too crowded, a new electron can’t find a place to sit. This is called “electron-electron repulsion.” It is like trying to squeeze into a crowded bus. Even if the bus driver wants you there, the other passengers might push you back! These small details make the electron affinity trend a very deep and interesting topic to study in school.
Comparing Electron Affinity and Electronegativity
Many people get the electron affinity trend confused with something called electronegativity. They are cousins, but they are not the same! Electron affinity is about a single atom grabbing an electron. Electronegativity is about how well an atom pulls on electrons when it is already bonded to another atom. Think of electron affinity as a solo sport, while electronegativity is more like a team sport. Both follow similar patterns on the periodic table, but they describe different moments in an atom’s life. Knowing the difference makes you a real chemistry pro!
Fun Facts About Atomic Energy
Did you know that Chlorine has the highest electron affinity of all the elements? Even though Fluorine is smaller, Chlorine has just the right amount of space to welcome a new electron without it being too crowded. This makes it a superstar in the electron affinity trend world. Another fun fact is that some atoms actually need you to force an electron onto them using extra energy. This shows that every element has its own unique “mood.” Exploring these facts makes science feel much more like a story than just a bunch of numbers.
Data Summary of Key Elements
To help you visualize the electron affinity trend, let’s look at a simple table. This table shows how the energy changes for some common elements. Remember, a higher negative number often means a stronger “hunger” for electrons in science charts.
| Element Name | Symbol | Trend Direction | Relative Affinity |
| Fluorine | F | Top Right | Very High |
| Chlorine | Cl | Top Right | Highest |
| Lithium | Li | Left Side | Low |
| Sodium | Na | Left Side | Low |
| Neon | Ne | Far Right | Almost Zero |
| Oxygen | O | Right Side | High |
Export to Sheets
Frequently Asked Questions
What is the general electron affinity trend? The general rule is that it increases as you move from left to right across a row and decreases as you move down a column.
Which element has the highest electron affinity? Chlorine actually holds the record for the highest electron affinity because it has the perfect size and space for an extra electron.
Why do Noble Gases have low electron affinity? Noble Gases have full outer shells. They are already stable and do not want to add more electrons, so they don’t release energy.
Is electron affinity the same as ionization energy? No, they are opposites. Ionization energy is the energy needed to take an electron away, while electron affinity is the energy released when adding one.
Does atom size affect the electron affinity trend? Yes! Smaller atoms usually have a stronger pull from the nucleus, which makes their electron affinity higher compared to larger atoms.
Can electron affinity be positive? Yes, for some elements that really do not want an electron, you have to use energy to force one in, making the value positive.
Conclusion: Mastering the Trend
We have traveled across the periodic table and seen how the electron affinity trend shapes our world. From the greedy Halogens to the peaceful Noble Gases, these patterns help us make sense of the tiny atoms that build everything we see. Understanding this trend isn’t just for passing a test; it’s about seeing the “energy language” of nature. Next time you see a piece of metal or breathe in oxygen, remember the tiny energy tug-of-war happening inside. Keep asking questions and exploring, because science is a never-ending adventure! Did this guide help you understand atoms better? Let us know!